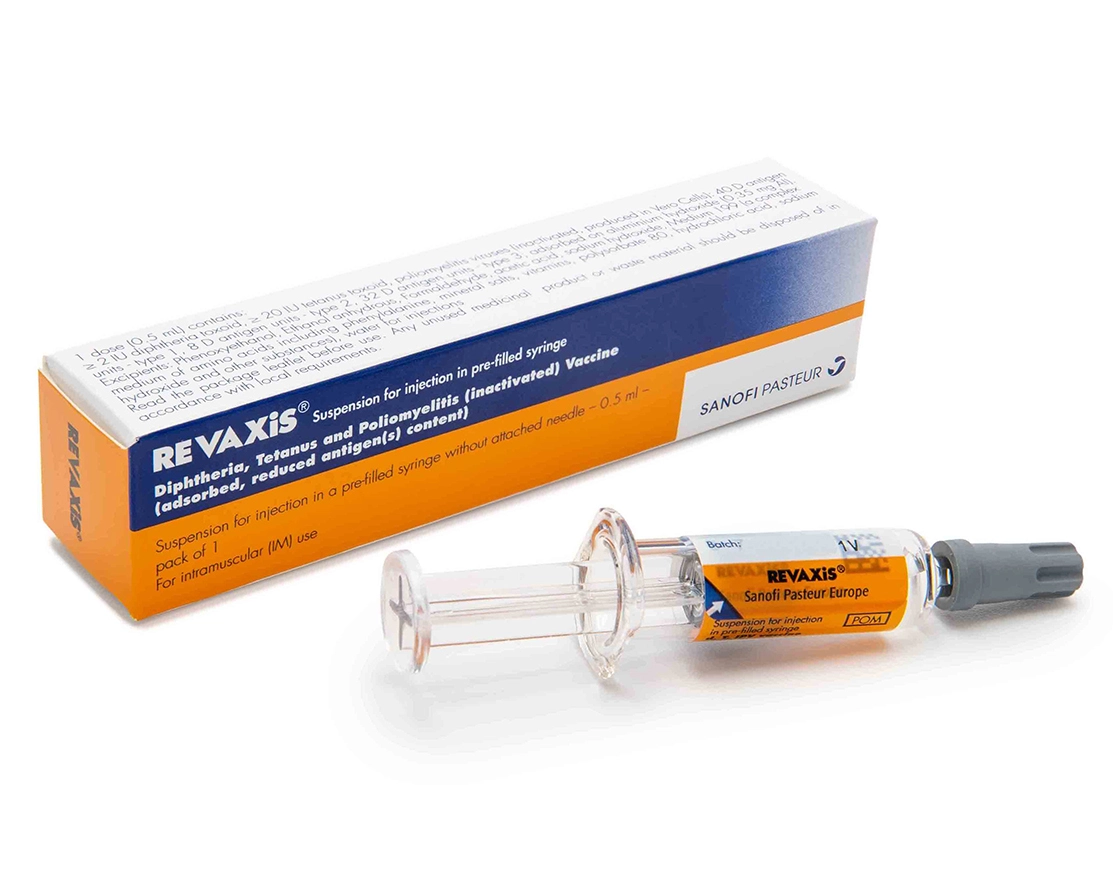
REVAXIS® Diphtheria, Tetanus & Poliomyelitis (inactivated) Vaccine (absorbed, reduced antigen(s) content) - Sanofi Campus

News - The Committee for Medicinal Products for Human Use at the EMA Recommends Authorisation of the Protein-Based COVID-19 Booster Vaccine VidPrevtyn Beta from Sanofi Pasteur - Paul-Ehrlich-Institut

GlaxoSmithKline and Sanofi report strong results in trials of Covid vaccine | GlaxoSmithKline | The Guardian
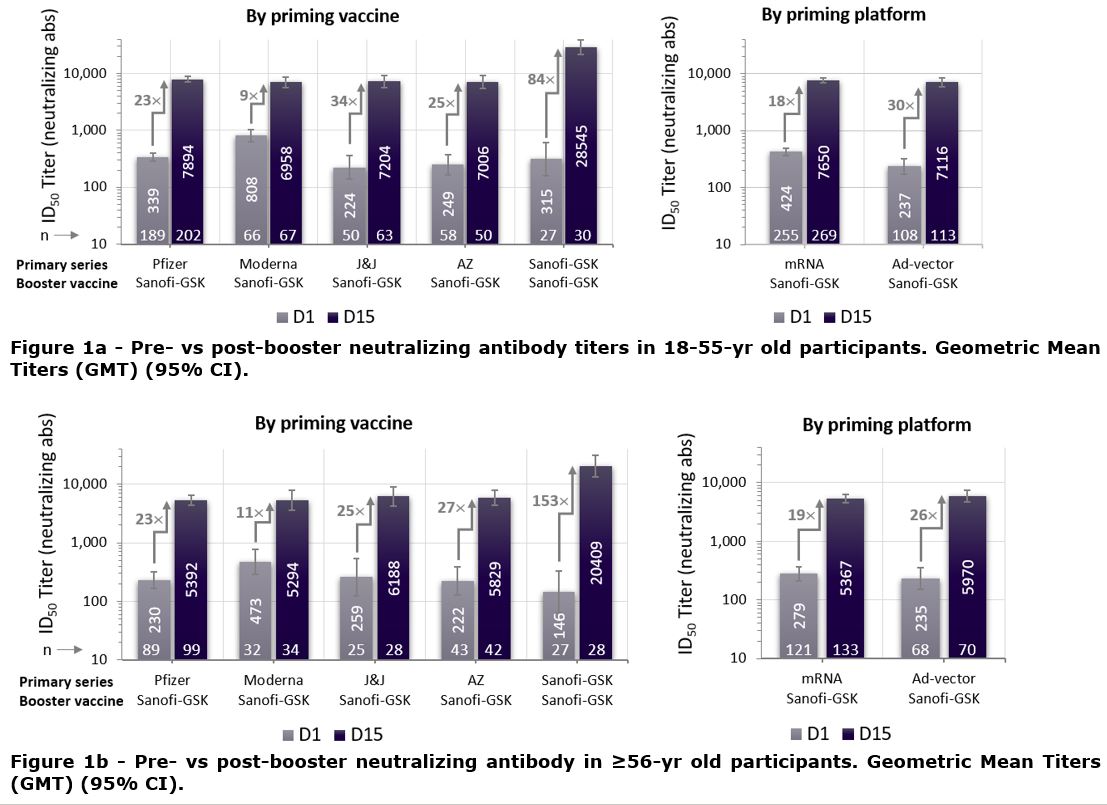
The immunogenicity and safety of the adjuvanted recombinant protein vaccine Sanofi/GSK-D614 or -B.1.351 administered as a heterologous booster dose